New clinical trials‘ online tool goes live
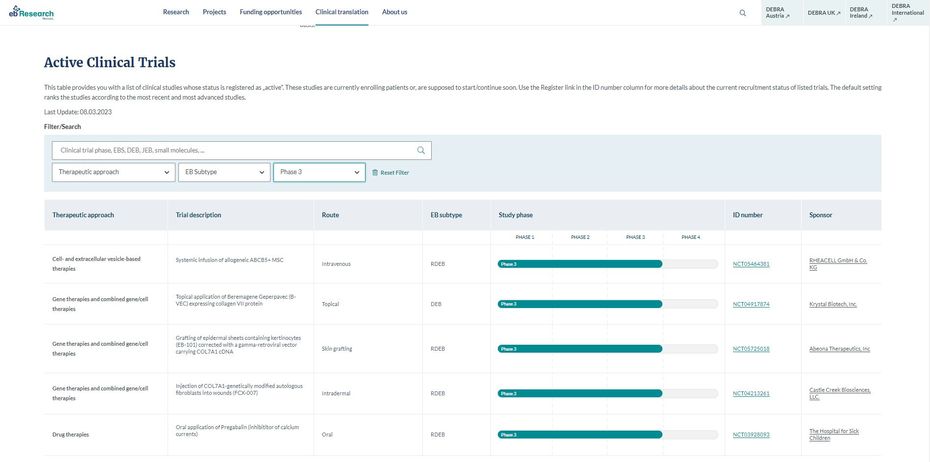
EB Clinical Trials modul
The approval of Amryt Pharma’s Filsuvez® - the world's first EB drug - in 2022 boosts other clinical trials for EB. The international trial registries show over 40 active clinical trials for EB worldwide. EB Resnet and EB Clinet have created a new online tool to make it easier to keep track of them.
The main aim of the tool is to provide a database with a pooled overview of all studies relevant to EB. It combines studies from the following registries: Clinicaltrials.gov, the EU Clinical Trials Registry, the International Clinical Trials Registry Platform (ICTRP), and the UMIN Clinical Trials Registry.
It will be available on EB Resnet’s and EB Clinet’s websites from mid-April 2023. You will find all registered ongoing and completed intervention and observational studies for EB. The EB Resnet team and the EB Clinet team will take care of the constant updating of the international database.
The database assigns the studies to the respective therapeutic approach, contains a brief description, and offers further information at a glance, such as the administration route, the EB subtype, the study phase, and the status. The database's filter functions help you refine your search by. In addition, a search function allows you to search specifically by keyword or, for example, for a specific sponsor.
You can click on the links to the respective study register and the study sponsor to obtain more detailed information. If you have specific questions about the studies, please get in touch with the respective study centre directly.
We hope the new tool will be helpful. Please feel free to communicate this to the EB community.
Note that we cannot guarantee the completeness of the lists, even though they are updated regularly. We are happy to support the maintenance of the database - information about missing studies or updates is very welcome. Thank you very much for your support.